MultiDose
The Easy, Accurate & Versatile Radiopharmaceutical
Dispenser for Syringes & Vials

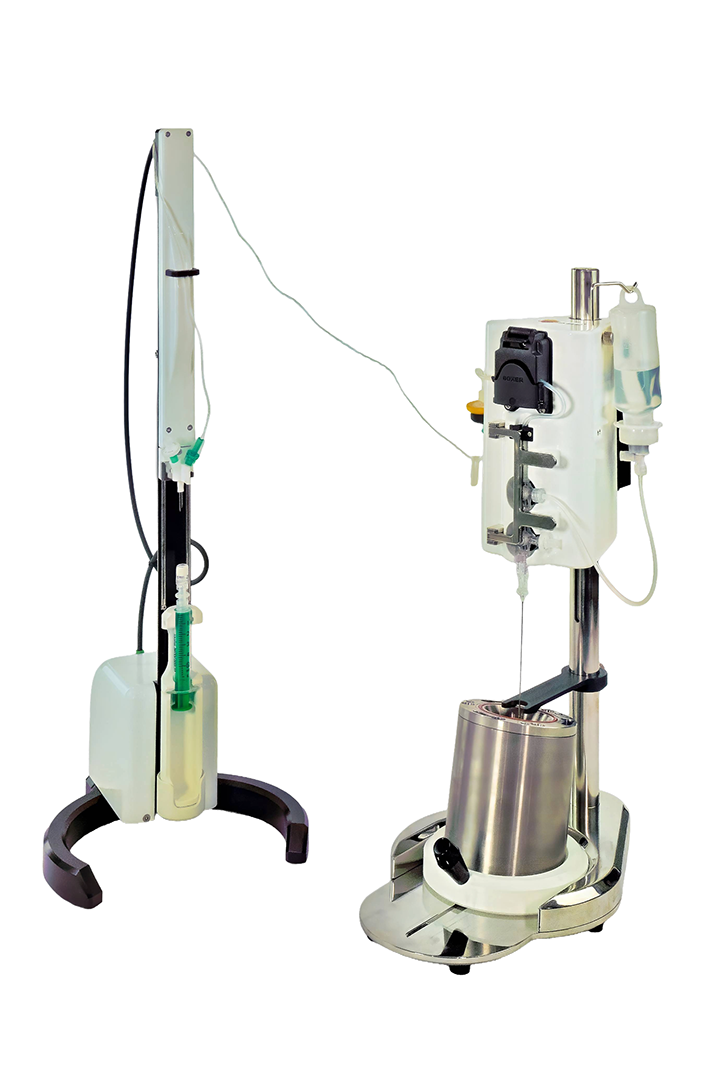
The MultiDose is a compact easy to use radiopharmaceutical dispenser for dispensing from a shielded bulk product vial into both vials and syringes.
It uses a disposable sterile stopcock assembly in combination with a peristaltic pump to accurately and quickly dispense doses. The bulk product vial can be automatically diluted before dispensing and also on-line dilution of the dispensed dose is possible.
The MultiDose has an intuitive and easy to user interface with built in communications to read a dose calibrator and integrated label printer.
DOWNLOAD BROCHURE
Key features
Intuitive & Easy to Use

21 CFR Part 11 & GMP compliant
Recording of all process variables and report generation
Reliability
Externally located PLC control box ensures all electronics are free from radiation damage
Label Printer
included for printing of syringe and vial labels pre and post dispensing
Chemical compatibility
All components compatible with vaporised H2O2
Externally Located Electronics
All key electronics are located external of the dispensing hotcell to ensure high radiation resistance
Dimensions (WxDxH)
Dispenser: 17.5 x 29.9 x 47.2 cm
Calibrator Lift: 20 x 21 x 50 cm

Dispensing Accuracy
± 5%
Minimum dispensing volume
50 μL
Disposable Cassette
Sterile disposable cassette manufactured by IPHASE
Shielded & Angled Bulk Product Vial
Angled orientation allows for maximum product recover with inserts available for 10 to 30ml vial sizes
Dispensing into Vials or Syringes
- 10, 15, 25 & 30 mL closed vials
- 5 or 10mL syringes (syringe fitted with injection site for closed syringe filling)
Dispensing Options
Dispense either by volume or activity with or without terminal sterile filtration
Automated Piercing
Automatic piercing of the bulk product vial and receiving vial (vented vial filling) or receiving syringe with all filling processes carried out while vial/syringe is lowered into the dose calibrators chamber
Integrated Dose Calibrator Reading
Integrated reading of compatible Dose Calibrator (user supplied). Currently suitable for use with:
- Capintec CRC-15
- Capintec CRC-25
- Capintec CRC-55
For other dose calibrators please contact IPHASE.

MultiDose Dispensing Process
Key steps for dispensing
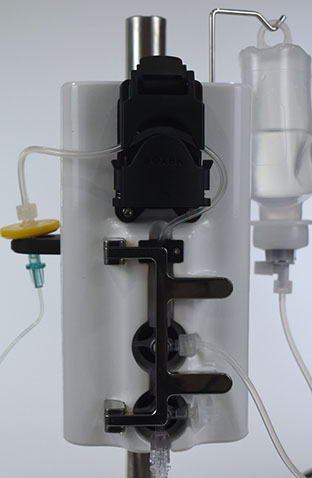
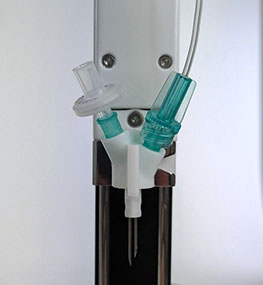
1: Cassette Assembly & Installation
Install disposable cassette onto dispenser with magnetic latch to lock into place and slide needle piercing clip onto calibrator lift
2: Record bulk product details
Enter bulk product vial details into software (Compound, Isotope, Batch & Volume)
and dose calibrator records product Activity & Time
2.jpg?crc=244818912)
2.jpg?crc=244818912)
3: Calibrate & Prime
- Dispenser automatically lowers to pierce bulk product vial.
- Attach calibration syringe or vial onto the calibrator lift
- Dispenser will automatically pierce the receiving vial/syringe and then lower into the dose calibrators chamber
- MultiDose will then dispense a small dose for activity concentration calculation and prime the stopcock/tube set.
- Once complete, the calibrator lift is raised and the vial/syringe automatically unpierced and the calibration vial/syringe can then be removed.

4: Dispense
- Install vial or syringe to dispense into on the calibrator lift
- Import and select dispensing data from "Dispensing List" or manually enter dispensing details
- Calibrator lift automatically pierces the vial/syringe and lowers into the dose calibrators chamber and dispensing starts

5: Completion of Dispensing
Upon completion of dispensing measured activity will be recorded and a summary of the dispensing details will be recorded and logged.
2.jpg?crc=244818912)
6: Label Printed
Print and attach automatically printed label to syringe/syringe shield or vial/vial pot. Pre-dispensing labels can also be printed

User Interface
Intuitive & Easy to Use
2.jpg?crc=244818912)
2.jpg?crc=244818912)
21 CFR Part 11 & GMP compliant
- Recording of all process variables and report generation.
- Multiple user access levels to maximise security and meet your regulatory needs.
- Data logging, reporting and audit trail capabilities included
- Record and store immutable dispensing batch information that can be viewed from within the software or stored as a pdf batch report.
- Audit trail records kept tracking device operation, user inputs, changes to set up parameters and more.

